WASHINGTON (AP) — The company responsible for a global recall of sleep apnea machines will be barred from resuming production at U.S. facilities until it meets a number of safety requirements, under a long-awaited settlement announced Tuesday by federal officials.
Philips will be required to overhaul its manufacturing and quality control systems and hire independent experts to vet the changes, according to a court order announced by the U.S. Department of Justice. The company must also continue to replace, repair or provide refunds to all U.S. customers who got the defective devices, the department said.
The action is a major step toward resolving one of the biggest medical device recalls in history, which has dragged on for nearly three years.
Most of the devices recalled are continuous positive airway pressure, or CPAP, machines. They force air through a mask to keep mouth and nasal passageways open during sleep. Left untreated, sleep apnea can lead to dangerous drowsiness and increased risk of heart attack.
 'Constantly learning' Imanaga off to impressive start with the Chicago Cubs
'Constantly learning' Imanaga off to impressive start with the Chicago Cubs War a real threat and Europe not ready, warns Poland's PM
War a real threat and Europe not ready, warns Poland's PM Boeing CEO Dave Calhoun to step down in shakeup amid safety crisis
Boeing CEO Dave Calhoun to step down in shakeup amid safety crisis Pence says he will not endorse former boss Trump in 2024 US election
Pence says he will not endorse former boss Trump in 2024 US election Ship that caused Baltimore bridge collapse has been refloated
Ship that caused Baltimore bridge collapse has been refloatedJon Wysocki dead at 53: Staind drummer passes away
Customs seize $20m meth and MDMA in two bags at Auckland Airport
 A Customs NZ photo showing 25.71kg of MDMA (ecstasy) stashed in a suitcase and seized at Auckland In ...[Detailed]
A Customs NZ photo showing 25.71kg of MDMA (ecstasy) stashed in a suitcase and seized at Auckland In ...[Detailed]Literacy, numeracy tests as NCEA requirement 'detrimental' to learners
 File pic Photo: 123RF ...[Detailed]
File pic Photo: 123RF ...[Detailed]Doctors who visited Gaza speak of 'atrocities', collapsing healthcare
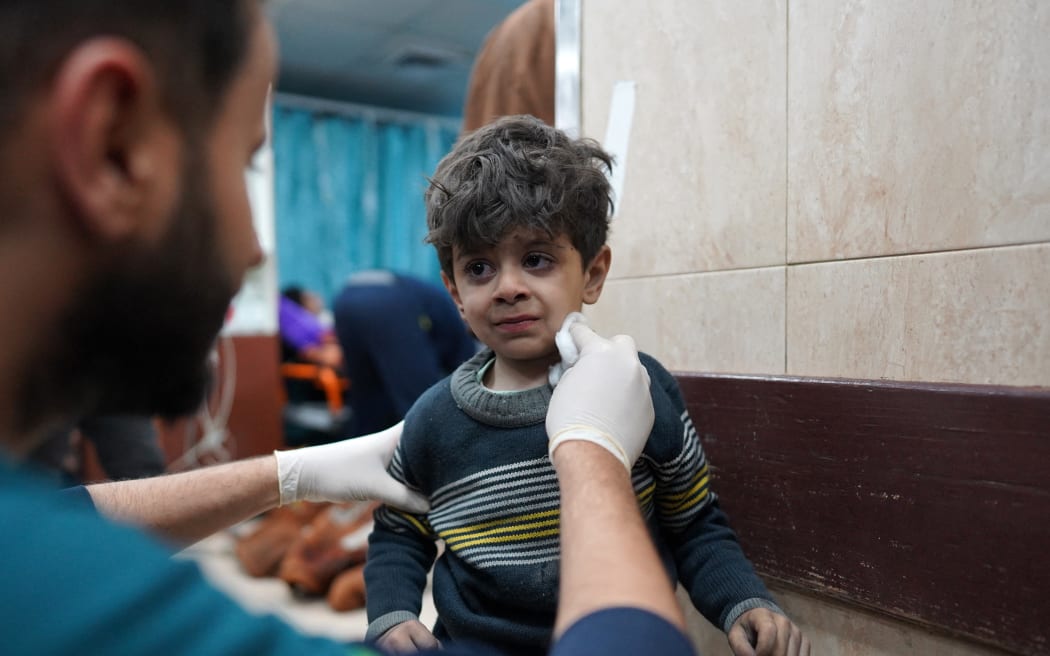 By Josie Kao, ReutersA Palestinian child receives medical attention at the Al-Aqsa Martyrs Hospital ...[Detailed]
By Josie Kao, ReutersA Palestinian child receives medical attention at the Al-Aqsa Martyrs Hospital ...[Detailed]Pope trip to Luxembourg, Belgium confirmed for September, 2 weeks after challenging Asia visit
 ROME (AP) — Pope Francis has added a stop in Luxembourg to a trip to Belgium in September, a three-d ...[Detailed]
ROME (AP) — Pope Francis has added a stop in Luxembourg to a trip to Belgium in September, a three-d ...[Detailed]Fashion designer Kate Sylvester to close the business after three decades
 File photo. Fashion designer Kate Sylvester. Photo: Kate Sylvester ...[Detailed]
File photo. Fashion designer Kate Sylvester. Photo: Kate Sylvester ...[Detailed]Whale euthanised after stranding near Christchurch
 Whale stranded at Banks Peninsula on 20 March 2024. Photo: Facebook / Project Jonah ...[Detailed]
Whale stranded at Banks Peninsula on 20 March 2024. Photo: Facebook / Project Jonah ...[Detailed]Journalist and commentator Rod Oram dies after cycling accident
 Rod Oram, pictured in a RNZ studio in 2016. Photo: RNZ/Dru Faulkner ...[Detailed]
Rod Oram, pictured in a RNZ studio in 2016. Photo: RNZ/Dru Faulkner ...[Detailed]Siblings trying to make US water polo teams for Paris Olympics
 WALNUT, Calif. (AP) — Chase Dodd started swimming when he was just a kid. Once he began playing wate ...[Detailed]
WALNUT, Calif. (AP) — Chase Dodd started swimming when he was just a kid. Once he began playing wate ...[Detailed]Old commuter carriages blighting Taumarunui scrapped after years of decay
 Twenty-seven of the 53 Auckland train carriages dumped in Taumarunui will be scrapped over the next ...[Detailed]
Twenty-seven of the 53 Auckland train carriages dumped in Taumarunui will be scrapped over the next ...[Detailed]Kate Hudson hits the stage to debut songs from her new album Glorious at star
Movement of pilot's seat a focus of probe into LATAM Boeing flight, report says
